
Covalent Modifiers: A Chemical Perspective on the Reactivity of α,β-Unsaturated Carbonyls with Thiols via Hetero-Michael Addition Reactions | Journal of Medicinal Chemistry
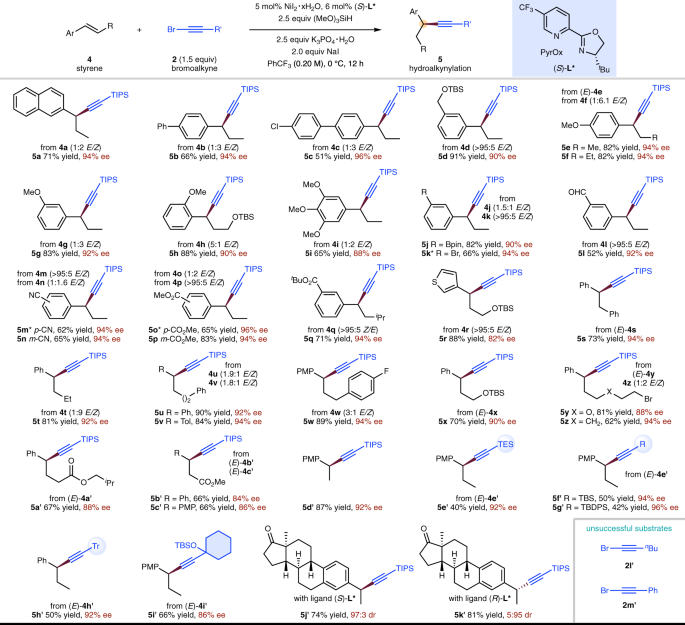
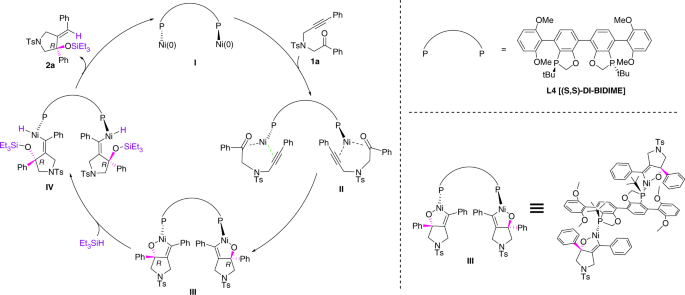
Nickel-catalysed migratory hydroalkynylation and enantioselective hydroalkynylation of olefins with bromoalkynes | Nature Communications

Nickel-catalysed migratory hydroalkynylation and enantioselective hydroalkynylation of olefins with bromoalkynes | Nature Communications

Facile Synthesis of Chiral Arylamines, Alkylamines and Amides by Enantioselective NiH‐Catalyzed Hydroamination - Meng - 2021 - Angewandte Chemie International Edition - Wiley Online Library

A biomimetic SH2 cross-coupling mechanism for quaternary sp3-carbon formation. - Abstract - Europe PMC

C/Meo Collective Women's Crossover Front Knit Wrap Sweater Top, Grey Sculpt, XS at Amazon Women's Clothing store

Development and Mechanistic Studies of (E)-Selective Isomerization/Tandem Hydroarylation Reactions of Alkenes with a Nickel(0)/Phosphine Catalyst | ACS Catalysis
Pyrrolidines and piperidines bearing chiral tertiary alcohols by nickel-catalyzed enantioselective reductive cyclization of N-alkynones | Communications Chemistry

Nickel-catalysed migratory hydroalkynylation and enantioselective hydroalkynylation of olefins with bromoalkynes. - Abstract - Europe PMC

Regio- and enantioselective remote hydroarylation using a ligand-relay strategy | Nature Communications

Facile Synthesis of Chiral Arylamines, Alkylamines and Amides by Enantioselective NiH‐Catalyzed Hydroamination - Meng - 2021 - Angewandte Chemie - Wiley Online Library

Low-Valent, High-Spin Chromium-Catalyzed Cleavage of Aromatic Carbon–Nitrogen Bonds at Room Temperature: A Combined Experimental and Theoretical Study | Journal of the American Chemical Society

Facile Synthesis of Chiral Arylamines, Alkylamines and Amides by Enantioselective NiH‐Catalyzed Hydroamination - Meng - 2021 - Angewandte Chemie - Wiley Online Library

Visible Light Photocatalysis: Applications and New Disconnections in the Synthesis of Pharmaceutical Agents | Organic Process Research & Development

Covalent Modifiers: A Chemical Perspective on the Reactivity of α,β-Unsaturated Carbonyls with Thiols via Hetero-Michael Addition Reactions. - Abstract - Europe PMC








